Background
Limited data are available on the clinical activity of venetoclax (ven) in mantle cell lymphoma (MCL). In 2 small retrospective studies of patients (pts) with MCL previously treated with BTK inhibitors (BTKi), ven resulted in overall response rates (ORRs) of 50-53% and median overall survival (OS) of 9-14 months (Eyre et al, Haematologica 2019; Zhao et al, Am J Hematol 2020). We sought to report the clinical activity of ven and identify factors associated with outcomes in a larger cohort of pts.
Methods
We included pts with relapsed MCL from 12 US medical centers treated with ven alone or in combination with an antiCD20 monoclonal antibody (mAb) or a BTKi. Response was determined by the local investigator. We defined OS as time from start of ven to death. Pts not experiencing an event were censored at their last known follow up. OS was determined using the Kaplan-Meier method, and univariable (UVA) and multivariable (MVA) models were developed to identify predictors of response and OS.
Results
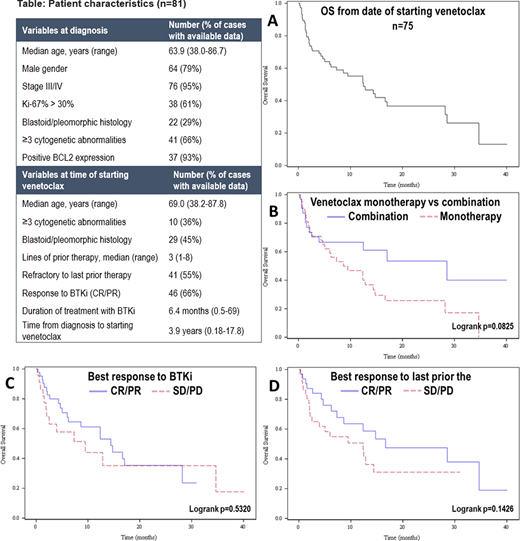
Eighty-one pts were included. Pt characteristics (Table) at diagnosis (dx) were: median age 64 years (yrs) (range 38-87), male 79%, stage III/IV 95%, Ki67 >30% in 61% (available n=62), blastoid/pleomorphic histology 29% (available n=75) and ≥3 cytogenetic abnormalities 34% (available n=62). BCL2 expression was present in 93% (available n=40). Frontline treatment (tx) included intensive chemotherapy [defined as including high-dose cytarabine and/or autologous transplant (ASCT) in first remission] in 52% with ASCT in first remission in 32%. Median number of therapies prior to ven was 3 (range 1-8) including antiCD20 mAb 99%, alkylator 93%, BTKi 91%, anthracycline 58%, cytarabine 56%, and lenalidomide 37%. 55% were refractory (defined as stable (SD) or progressive disease (PD)) to last tx prior to ven. ORR to BTKi prior to ven (n=70) was 66% (complete response (CR) 20%) with median duration of tx of 6.4 months (range 0.5-69). BTKi was stopped due to PD 82% and toxicity 18%. Median time from dx to start of ven was 3.9 yrs (range 0.2-17.8). Ven was given as monotherapy in 62% (n=50) and in combination with BTKi 20% (n=16), antiCD20 mAb 14% (n=11), or other 5% (n=4). Ten pts treated with ven in combination with BTKi received prior tx with BTKi with ORR to BTKi of 40% (all PR). Ven highest dose received was 20-100 mg in 12% (n=9), 200 mg 11% (n=8), 400 mg 61% (n=46), and 800 mg 17% (n=13). Median duration of tx with ven was 2.8 months (range 0.1-30). The best response to ven was CR 18%, partial response (PR) 24%, SD 11%, and PD 47% with ORR of 42%; 19 pts (23%) did not have available data for response. ORR was not significantly different with ven monotherapy 36% (13/36) vs ven + antiCD20 mAb 56% (5/9) vs ven + BTKi 43% (6/14) (p=.559), or with ven monotherapy 36% vs combination therapy 50% (13/26) (p=.274). Ven was stopped due to PD 69%, toxicity 9%, allogeneic transplant 3% or other reasons 19%. Laboratory tumor lysis syndrome (TLS) occurred in 10 pts (12%) including 3 (3.7%) with clinical TLS. 38 pts received post-ven tx with median time from stopping ven to next tx of 0.24 months (range 0-2.2); for the 33 pts with available data, the best response to post-ven tx was CR 24%, PR 27%, SD 9% and PD 39% with ORR of 51%. For the 75 pts with available data, median OS was 12.5 months (95% CI 6-17) with 3-year OS of 13.1% (95% CI 1.4-38.0%) (Figure A). Median OS was numerically longer with ven combination (28.7 months, 95% CI 4-not reached) vs monotherapy (8.6 months, 95% CI 4.7-14.4), p=.0825 (Figure B). Median OS did not significantly differ based on response (CR/PR vs SD/PD) to prior tx with BTKi (14.4 vs 9.5 months, p=.53, Figure C) or last tx prior to ven (16.7 vs 12.4 months, p=.14, Figure D). In MVA for response, achieving CR/PR with BTKi prior to ven (vs SD/PD) was associated with response to ven (odds ratio 3.48, 95% CI 1.01-12.05, p=.049). In MVA for OS, ven combination vs monotherapy (HR 0.13, 95% CI 0.03-0.53; p=.006), longer time from dx to start of ven (>4 vs ≤4 yrs) (HR 0.08, 95% CI 0.02-0.36; p=.001), and higher dose of ven (≥400 mg vs <400) (HR 0.17, 95% CI 0.04-0.72; p=.016) were associated with superior OS, while longer duration of tx with BTKi (>6 vs ≤6 months) (HR 3.47, 95% CI 1.10-10.99; p=.038) was associated with inferior OS.
Conclusions
In these high-risk and heavily pretreated pts with MCL, ven resulted in low response rate and poor OS. Ven may have a better role in MCL in earlier lines of therapy and when combined with other agents such as BTKi and/or antiCD20 mAbs.
Kamdar:Roche: Research Funding. Greenwell:Lymphoma Research Foundation: Research Funding; Acrotech Biopharma LLC, Kyowa Kirin: Consultancy. Hess:ADC Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; BMS, AstraZeneca: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Portell:Acerta/AstraZeneca: Research Funding; Kite: Consultancy, Research Funding; Amgen: Consultancy; Janssen: Consultancy; Pharmacyclics: Consultancy; Bayer: Consultancy; BeiGene: Consultancy, Research Funding; AbbVie: Research Funding; TG Therapeutics: Research Funding; Infinity: Research Funding; Roche/Genentech: Consultancy, Research Funding; Xencor: Research Funding. Goldsmith:Wugen Inc.: Consultancy. Grover:Genentech: Research Funding; Tessa: Consultancy. Riedell:Morphosys: Research Funding; Celgene/Bristol-Myers Squibb Company: Honoraria, Research Funding; Verastem Oncology: Honoraria; Novartis: Consultancy, Honoraria, Research Funding; Kite Pharmaceuticals/Gilead: Honoraria, Research Funding; Bayer: Honoraria; Karyopharm Therapeutics: Honoraria. Karmali:BeiGene: Speakers Bureau; AstraZeneca: Speakers Bureau; Karyopharm: Honoraria; Takeda: Research Funding; Gilead/Kite: Honoraria, Other, Research Funding, Speakers Bureau; BMS/Celgene/Juno: Honoraria, Other, Research Funding, Speakers Bureau. Kumar:AbbVie: Research Funding; Kite Pharmaceuticals: Honoraria, Other: Honoraria for Advisory Board; Astra Zeneca: Honoraria, Other: Honoraria for Advisory Board; Celgene: Honoraria, Other: Honoraria for Advisory Board; Seattle Genetics: Research Funding; Pharmacyclics: Research Funding; Celgene: Research Funding; Adaptive Biotechnologies,: Research Funding. Hill:Abbvie: Consultancy, Honoraria, Research Funding; Pharmacyclics: Consultancy, Honoraria, Research Funding; Beigene: Consultancy, Honoraria, Research Funding; AstraZenica: Consultancy, Honoraria, Research Funding; Kite, a Gilead Company: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria; BMS: Consultancy, Honoraria, Research Funding; Karyopharm: Consultancy, Honoraria, Research Funding; Takeda: Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Genentech: Consultancy, Honoraria, Research Funding. Kahl:AstraZeneca Pharmaceuticals LP: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene Corporation: Consultancy; AbbVie: Consultancy; Genentech: Consultancy; Pharmacyclics LLC: Consultancy; Roche Laboratories Inc: Consultancy; BeiGene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Acerta: Consultancy, Research Funding; ADC Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Cohen:Genentech, BMS, Novartis, LAM, BioInvent, LRF, ASH, Astra Zeneca, Seattle Genetics: Research Funding; Janssen, Adicet, Astra Zeneca, Genentech, Aptitude Health, Cellectar, Kite/Gilead, Loxo: Consultancy.
Venetoclax is not licensed for use in mantle cell lymphoma
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal